 
14.5: Periodic Trends: Atomic Size, Ionization Energy, and Metallic Character Certain properties-notably atomic radius, ionization energy, electron affinity and metallic character-can be qualitatively understood by the positions of the elements on the periodic table.The table is divided into metals, nonmetals, and metalloids, each with distinct properties. Elements in the same group share similar characteristics, like reactivity. These variables allowed Mendeleev to place each element in a certain row (called a period) and column (called a group). You can look up element names, symbols, atomic masses and more, or test your knowledge with a periodic table game. Electron configurations can be predicted by the position of an atom on the periodic table. The periodic table organizes elements into groups and periods based on their chemical and physical properties. The periodic table is arranged by atomic weight and valence electrons. PubChem Periodic Table of Elements is an interactive tool that lets you explore the properties and trends of chemical elements. 14.3: Electron Configurations and the Periodic Table (The Block Method) The arrangement of electrons in atoms is responsible for the shape of the periodic table.This page titled 5.2: Covalent Bonds and the Periodic Table is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by Anonymous. How to determine the number of valence electrons and draw Lewis structures for main group elements starting from the electron configuration. Point to Remember: Mostly for transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core. The atoms in group 6A make two covalent bonds. Thus the inner transition elements have valence electrons ranging from 3 to 16 (See above image of periodic table). Hund’s rule says that the lowest-energy arrangement of electrons is the one that places them in degenerate orbitals with parallel spins. Covalent bonds are formed by two atoms sharing electrons. 14.2: Electron Configurations Based on the Pauli principle and a knowledge of orbital energies obtained using hydrogen-like orbitals, it is possible to construct the periodic table by filling up the available orbitals beginning with the lowest-energy orbitals (the aufbau principle), which gives rise to a particular arrangement of electrons for each element (its electron configuration). 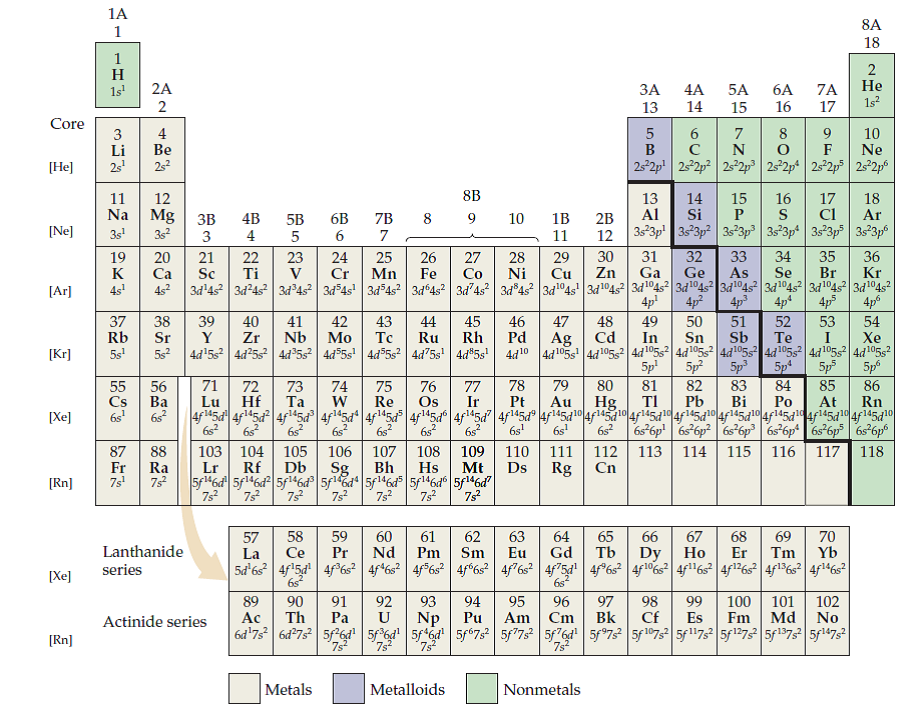
14.1: The Perioidic Table The chemical properties of elements is determined primarily by the number and distribution of valence electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
